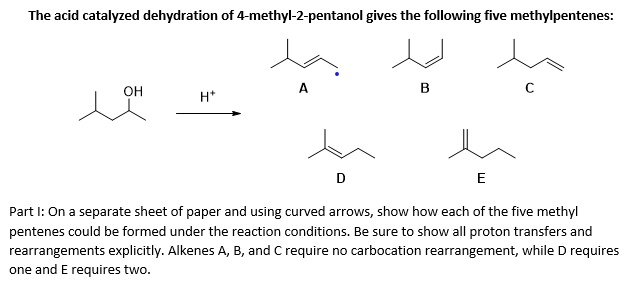
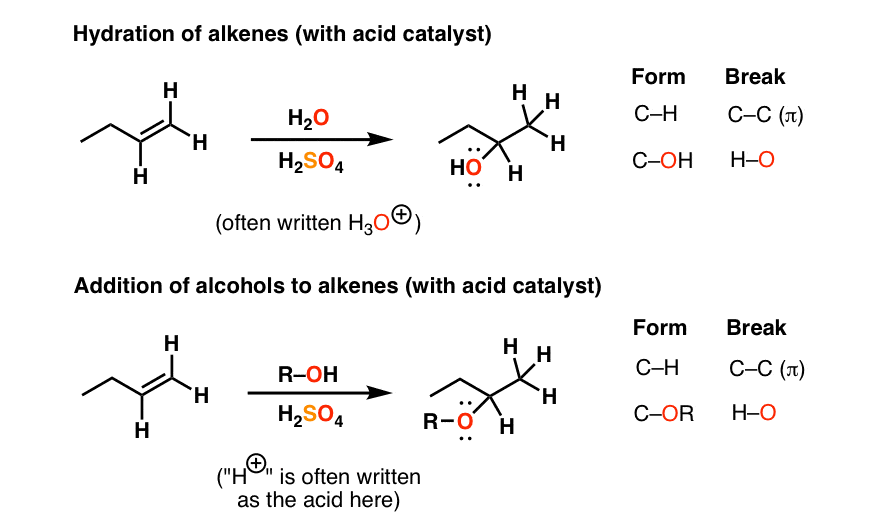
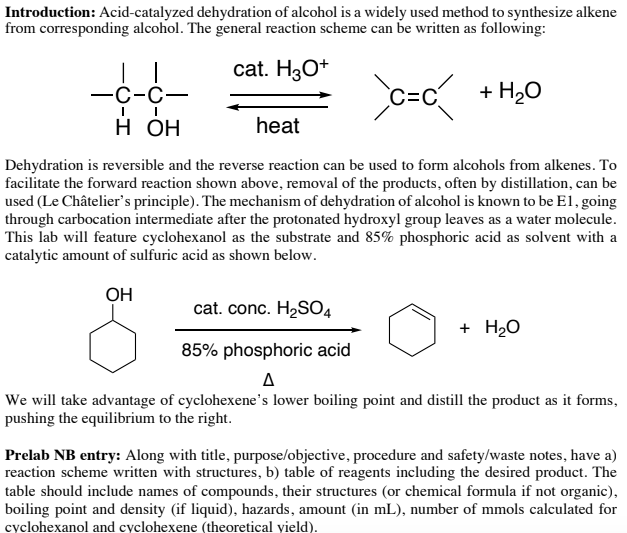
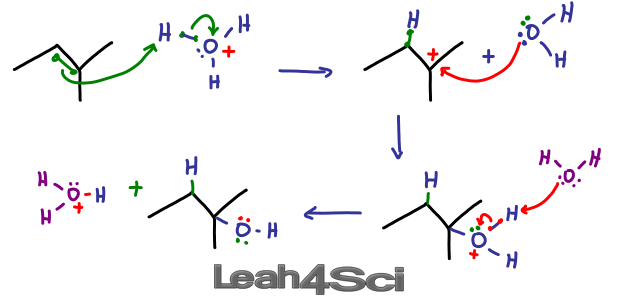
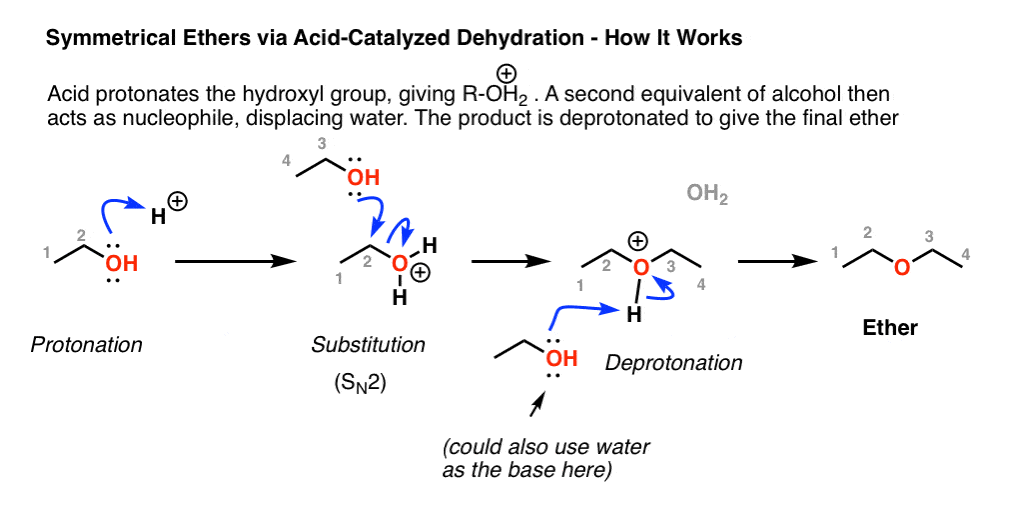
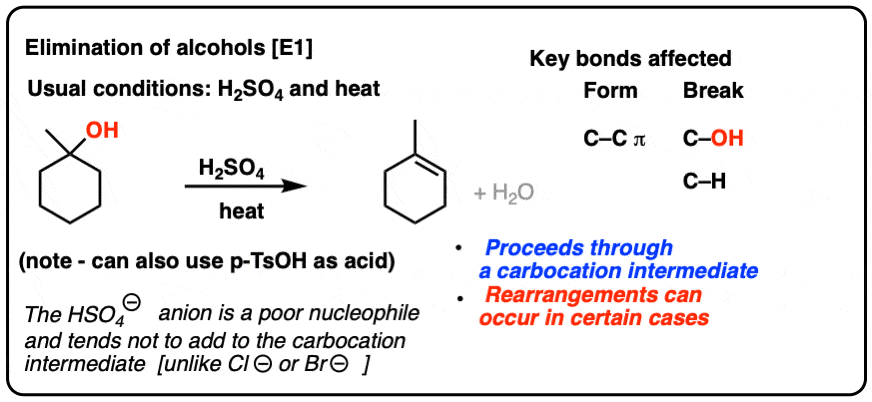
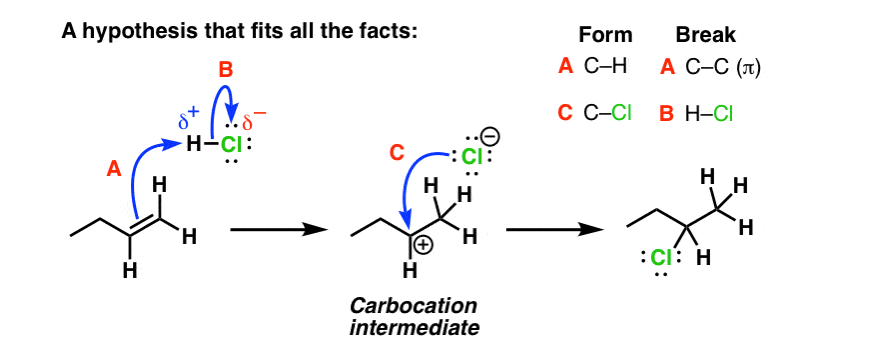
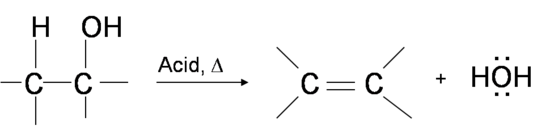Preparation of ethers by acid dehydration of secondary or tertiary alcohols is not a suitable method. Give reason.

organic chemistry - Yield of possible products from acid catalysed hydration of an alkene with a phenyl group - Chemistry Stack Exchange

In acid catalyzed dehydration of alcohol which of the following alkene is formed. The reaction may involve rearrangement.

What is the mechanism for the acid catalyzed dehydration of an alcohol to form an alkene? | Homework.Study.com

a) Write the mechanism of acid catalysed dehydration of ethanol to ethene.b) Between phenol and alcohol which is more acidic? Why?

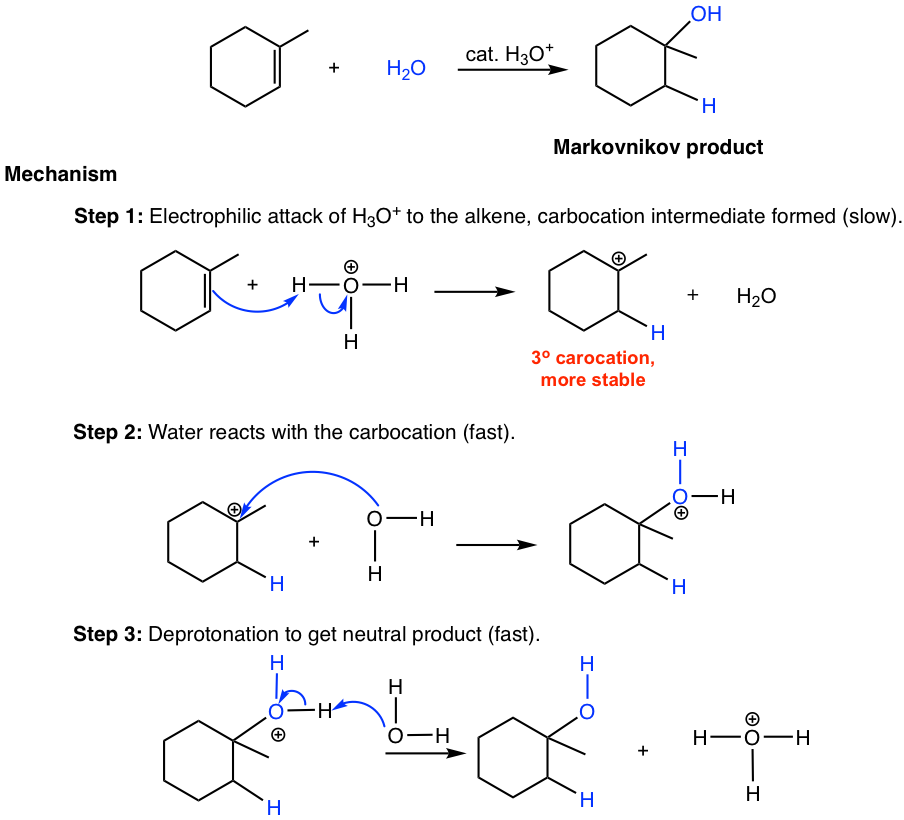
Predict the major product of acid catalysed dehydration:(i) 1 - methyl cyclohexanol(ii) Butan - 1 - ol